Note that in Fe(NO 3) 3, there are three nitrate ions, i.e., one iron atom, three nitrogen atoms, and nine oxygen atoms, in the formula unit of the compound. For example, iron(III) nitrate is Fe(NO 3) 3 sodium carbonate is Na 2CO 3 ammonium phosphate is (NH 4) 3PO 4 potassium permanganate is KMnO 4 and calcium phosphate is Ca 3(PO 4) 2. While these are the most common valences, the real behavior of electrons is less simple. These can be found in nature in both free and combined states. It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. The oxygen family, also called the chalcogens, consists of the elements found in Group 16 of the periodic table and is considered among the main group elements. The practical oxygen impurity minimum of 2 × 10 16 cm 3 is set by the 700 C decomposition temperature of GaN in ultra-high vacuum. You may assume that the valences of the elementsthe number of electrons with which an atom will bond or formare those that can be derived by looking at the groups (columns) of the periodic table. Group 16: General Properties and Reactions. 
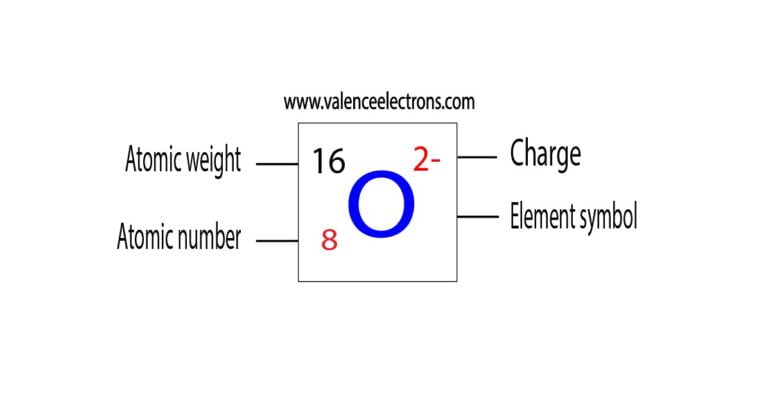
Just like the subscript to the right of the monoatomic anion tells how many atoms of the anions are there, the subscript to the right of the small bracket around a polyatomic anion tells how many polyatomic anions are there in the compound. A reduction in the oxygen concentration of one order of magnitude is observed when increasing the growth temperature by 60 C. If a subscript is needed for the anion, place the polyatomic ion within small brackets and write the subscript outside the bracket. The writing formula of compounds containing polyatomic ions is the same as writing the formula of a binary ionic compound, except that the polyatomic ions must remain intact as a unit. d Atomic EDS mapping showing the disordered elemental distribution in the B-site of the ABO3 structure. 'Direct observation of elemental fluctuation and oxygen octahedral distortion-dependent charge distribution in high entropy oxides,' Nature Communications, Nature, vol. The green and silver dots marked in c denotes the B-site atoms and oxygen atom, respectively. The polyatomic ion acts as a single unit, i.e., they are molecular ions. Lei Su & Huaixun Huyan & Abhishek Sarkar & Wenpei Gao & Xingxu Yan & Christopher Addiego & Robert Kruk & Horst Hahn & Xiaoqing Pan, 2022. Writing formulae of compounds containing polyatomic ions Table 1: Names of some of the common polyatomic ions Formula It is defined as being the charge that an atom would have if all bonds were ionic. Table 3.3.1 lists the formulas and names of some of the common polyatomic ions. Living on Mars requires the ability to synthesize chemicals that are essential for survival, such as oxygen, from local Martian resources. Element Oxygen (O), Group 16, Atomic Number 8, p-block, Mass 15.999.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
